Our research focuses on the post-translational modifications (PTMs) of proteins particularly ubiquitination, deamidation and phosphorylation, regulated by pathogenic bacteria. It is well understood that PTMs regulate host cell homeostasis and disease development such as cancer, inflammatory and infectious diseases. Many pathogenic bacteria target specific PTM pathways to establish infection; however, little is known about the significance and underlying mechanisms of these PTM pathways. Therefore, the aims of our research are (1) to characterize the various bacterial PTM systems and determine their target host proteins, and (2) to elucidate the underlying molecular mechanism of the PTM systems active during bacterial infection. We use biochemistry, chemical biology, and animal models to understand how pathogenic bacteria use PTMs of proteins. The main goal of our research is to develop new therapeutic tools that will target these PTMs for the treatment of serious human diseases, such as infectious disease and cancer.
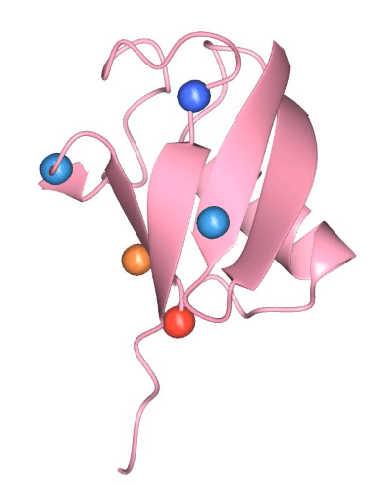
PTM of Ubiquitin
Ubiquitin is highly conserved small protein that covalently attached to the substrate protein. This post translational modification is called “ubiquitination”. Ubiquitin itself undergoes a variety of PTM, such as acetylation(light blue), phosphorylation(blue) and deamidation (red). Interestingly, Ubiquitin can be modified by other PTM during bacterial infection. We are interested in understanding how post-translationally modified ubiquitin modulate host ubiquitin system and the pathways that affect bacterial survival.
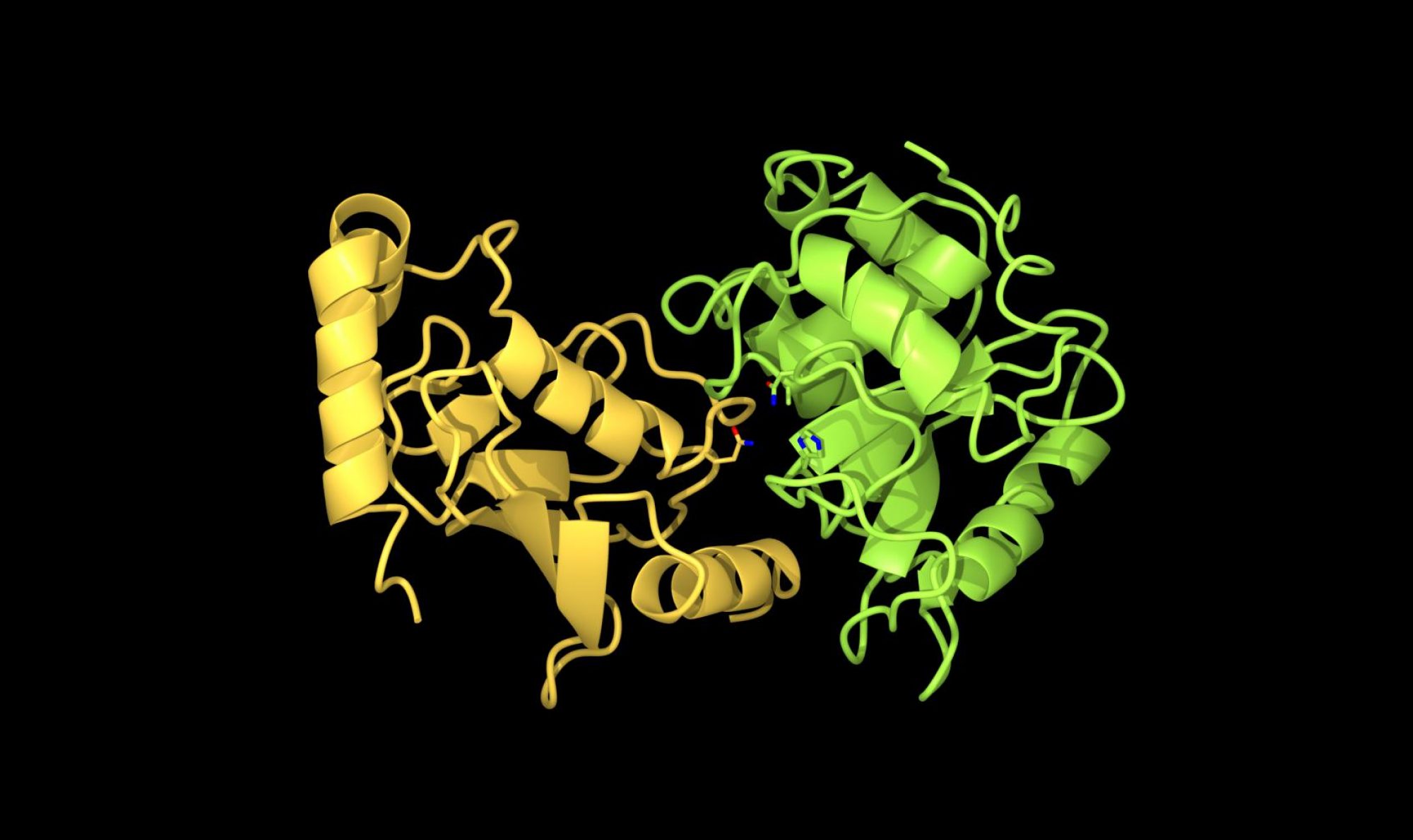
Bacterial ubiquitin ligases
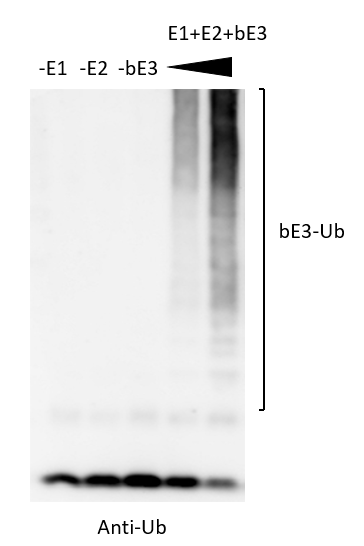
Ubiquitination regulates intracellular proteolysis and various cellular processes. Ubiquitination requires a cascade of three enzymatic (E1-E2-E3) reations. For a long time, people thought the ubiquitin system is conserved only in eukaryote. However, many pathogenic bacteria uses ubiquitin system for their own benefit. We are studying several bacterial ubiquitin ligases(E3) that interact and modulate the host’s ubiquitin-proteasome system during pathogenic bacterial infection. We are using chemical biology and biochemistry approaches to identify the target proteins, and thereby obtain new insights into the modulation of the host defense system.

Targeted Protein Degradation for drug discovery
Infectious diseases are an increasing problem worldwide (as we witness with the Coronavirus pandemic), while the emergence of multidrug-resistant bacteria is a major threat. Using Proteolysis targeting chimera technology (PROTAC), we are aim to develop new antibacterial and anticancer drugs. Artwork by M. T.

細菌と戦う私達のボディー
多くの抗生物質を手にした現代でも、我々は毎日細菌による感染症の大きな脅威にさらされています。私達の研究室では、細菌が体内に侵入し、感染を拡大する際に、我々の体がどのような感染防御策を持つかについて研究しています。さらにその成果を基に新しい感染症治療方法の開発を目指しています。 Artwork by A. N.
https://research.kyoto-u.ac.jp/academic-day/a2019/a2019-p016/